Article: Advanced Therapy Commercial Supply Chain Lessons for Clinical Trial Success
By AmerisourceBergen
A new way of thinking
An article published in Cell and Gene Therapy Insights[1] posed the following questions:
1. Is the established clinical flow - PhI to PhII to PhIII to commercial - applicable to advanced therapies?
2. Should the clinical supply chain be the same as the commercial supply chain for advanced therapies?
The insight argues that the answer to both these questions is “No”.
The 10+ years of development required to bring a traditional pharmaceutical to market is not seen in advanced therapies, with the target being closer to five years to move from first patient to commercial launch. In addition, traditional pharma changes drastically as it moves to commercial. This is because the supply chain is passed from the data driven, clinical trials paradigm into an integrated and outsourced supply chain. However, advanced therapies require time sensitive and temperature critical logistics platforms regardless of their development stage, meaning that the clinical and commercial logistics platforms only differ in terms of scale.
Therefore, for advanced therapies, a risk-based understanding is required that identifies Critical Logistics Attributes such as Chain of Identity and real-time temperature monitoring. This new way of thinking is summarized within five stages of development:
Stage 1: Envision
Traditionally, activities and planning for logistics, digital supply chain management and other enabling technologies are more limited in this stage. However, as advanced therapy developers acknowledge new early-phase complexities, an increasing number are doing more systems planning during Envision and including discussion of these early preparations in their IND filings.
Stage 2: Build
In the Build stage, activities and planning for logistics platforms, digital supply chain management and other enabling technologies affect this need for speed. While these systems may not yet need to scale out widely, they will need to support accelerated timelines as well as high expectations from clinical site investigators, quality teams, investors and regulatory experts.
Stage 3: Operate
In the Operate stage, advance work on logistics platforms, digital supply chain management and other enabling systems repeatedly demonstrates a return on investment. If the necessary foundation has been designed thoughtfully and planned in advance, this phase points the way towards a fully-developed vision of commercial success.
Stage 4: Optimize
In this stage, logistics systems, digital supply chain management and other enabling technologies should now be running in real time. Logistics is now a key concern, so a fully formed logistics platform is required. A documented, validated digital supply chain management system is also a requirement, and internal integrations between a full range of e-systems should now be underway.
Stage 5: Grow
Finally, at the Grow stage, certain aspects of the underlying systems and processes, such as manufacturing, are standardized and validated, so therefore are somewhat fixed. However, logistics platforms and digital supply chain management systems can continue to evolve within approved regulatory parameters – and be used as points of differentiation in an ever more competitive market.
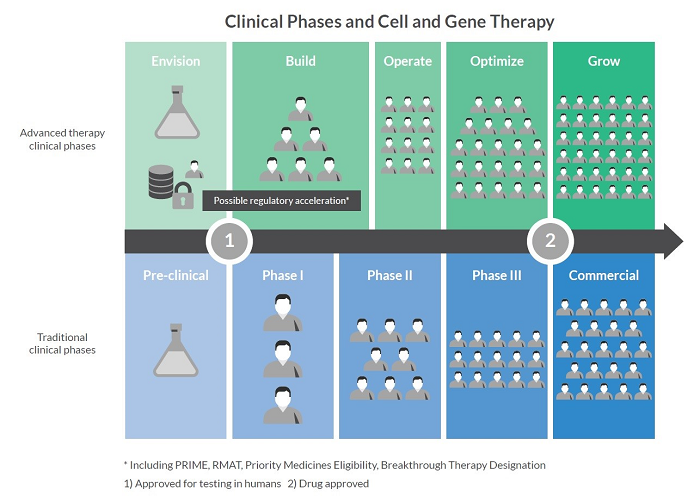
Ref: Commercial Lessons for Clinical Success, Cell & Gene Therapy Insights, Dec 2019
Essentially, advanced therapies require a new way of thinking if the industry is going to continue to grow. Sponsors need to align the manufacturing, clinical and logistics platforms if they want to scale smoothly, as well as using data to manage, monitor and control the journey of their therapies through a considerably complex supply chain.
An earlier version of this article was published in Cell & Gene
Therapy Insights co-authored by Simon Ellison, World Courier, Heidi Hagen and
Christophe Suchet, Vineti.
[1] Commercial Lessons for Clinical Success, Cell and Gene Therapy insights, Dec 2019. https://insights.bio/cell-and-gene-therapy-insights/journal/articles/commercial-lessons-for-clinical-success/



