6 questions with Kyle Noonan
By Kyle Noonan, PharmD, MS

Discussing payer perspectives of cell therapy and gene therapy treatment paradigms for an Inherited retinal disorder


What inspired this research?
Right now, most of the field looks at cell and gene therapy as one thing, when in fact, cell therapy and gene therapy are very different.
We have many gene therapies that have started to enter the market, but we also are starting to see the potential of some cell therapies in the pipeline that may be coming to market in future years. These treatments are completely different in the fact that gene therapy is a one-time treatment that's curative, but it only targets one gene. There might be diseases out there like certain retinal disorders where the same disease is caused by numerous genes. And that's where cell therapies could fill that gap because they're gene agnostic., so they could provide a cure to all patients, regardless of the specific gene mutation they have.
Since none of these cell therapies are on the market yet, there’s still a lot of uncertainties. But it's interesting to start parsing those apart because the research currently shows that the payers in the field just of cell and gene therapy think of them as one monolithic thing when it’s really not.
Was there a hypothesis that was confirmed through the research?
We expected the payers to view things very similarly. The caveat here is that we used a hypothetical narrative to describe an inherited retinal disorder and level set on what the implications would be when they make these decisions, in order to make it more of a real-world exercise that they were going through when trying to evaluate a therapy. Then we broke it out between a cell therapy paradigm and a gene therapy paradigm.
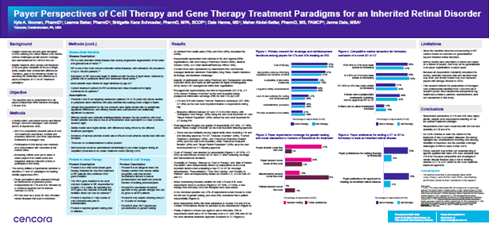
We were hoping that, without leading them too much, they would hone in on the fact that the gene therapy only treats one gene, which is only about 25 percent of the patients with retinitis pigmentosa, which was the narrative case used for this research. As we saw, they didn't really notice that the cell therapy could function as gene agnostic and treat all the patients’ retinitis pigmentosa, not just the 25 percent that have the most common gene mutation, which is what the model gene therapy was targeting in this research.
What are the key takeaways from your research?
The research was done through a survey during the Fall of 2023 to 32 U.S. payers from health systems and integrated delivery networks.
Most payers are currently looking at these very similarly. When we ask them different questions about the types of information they want to see for coverage and reimbursement decisions, they answered the same for both cell therapy and gene therapy treatment paradigms. But what's interesting is that when you start to talk about their preference to choose either a cell therapy or a gene therapy, they unanimously select gene therapy. 75 percent of payers would select gene therapy over cell therapy. Even though they still view them very similarly in the characteristics they want to know about these drugs, they still have a gravitation towards the gene therapy. And some of that could be because there's no cell therapy of this nature on the market yet.
The other big thing that we found from this research was once you start to get to the second therapy to market or later, payers are more prone to making formulary exclusions and starting to be choosy. One or two therapies market would get their attention, but past that second one, you might start to see exclusions where some payers are just going to only offer one or two of these therapies.
Why retina?
This disease state is progressive and severe. Most patients will reach legal blindness by age 40. There is a cellular-based therapy in development that would function agnostic of any different gene. With this disease, there are up to 70 different genetic mutations that can cause this same disease state. If you bring a cell therapy to market, you could treat all those patients or potentially treat refractory patients who didn't respond to gene therapy. Whereas gene therapy would only hit about 25 percent of patients, if it targeted the most common mutation.
Was there anything in the research that was surprising, that you didn't expect, that you found out?
Even though payers are still rating these therapies very similarly, they still had a strong gravitation to choose the gene therapy as if they had one choice. We don't quite know why. Maybe it's because they're just more familiar with it. It could be that there aren't any cell therapies of this type yet, so they just don't have a strong understanding and just sort of defaulted to gene therapy. We would've expected it to be a little more balanced
One of the downfalls of the research is we don't have any of these cell therapies out there, and they can't look at a real-world case yet. There are therapies in the pipeline, but that’s we framed it to be a hypothetical narrative.
What are the next steps from this research?
The next step would be to continue to track this over time. Maybe field something similar in a couple of years. It would be really interesting if we get similar therapies to market in this situation, to come back to them and start to understand these questions once they've had time with them in the real world. And even beyond that, just if we get any cellular therapies beyond CAR-T, because CAR-T doesn't really fit this model of what we were looking at, that would be a good cue to revisit this research.
Citations relevant to the content described herein are provided in the article linked here. Readers should review all available information related to the topics mentioned herein and rely on their own experience and expertise in making decisions related thereto.