Orphan drugs: trapped in the lock? A review of the lock system in the Netherlands
By HTA Quarterly
Whilst orphan diseases individually are rare, collectively they are not; population prevalence of rare diseases is put at 5% to 10%. Whilst developers look for high prices to cover their costs, can payers afford them? A review of the “lock” system in the Netherlands suggests maybe not.
In Europe, any disease affecting fewer than 5 people in 10,000 is considered orphan, and a subset of ultra-orphan diseases has been defined as those with a prevalence of ≤1 in 50,000 patients. Small population sizes present challenges to developing drugs to treat them; recruiting patients in sufficient numbers to conduct clinical trials is time-consuming and expensive, whilst limited patient numbers mean limited revenues in return. As a consequence, developing drugs for orphan diseases was historically unattractive so regulatory initiatives were introduced in 2000 in Europe to incentivize development, including lower fees and 10 years of market exclusivity after registration. These incentives have proved extremely successful as 19% of marketing authorization applications approved by the EMA in 2021 were for orphan conditions.
Developers often look for high prices as a result of the low patient numbers. However, whilst orphan diseases individually are rare, collectively, they are not; an analysis of the Orphanet database yielded a conservative, evidence-based estimate for the population prevalence of rare diseases of 3.5% to 5.9%. Consequently, the cost of treating orphan diseases as a whole is likely to be too high for payers to accept.
In the Netherlands, orphan drugs historically qualified for automatic reimbursement on registration as hospital drugs were not assessed. However, in 2015 the Ministry of Health and Welfare (MoH) piloted the “lock” system in reaction to increased pressure on hospital budgets and formalized it into law in 2018. As a result, orphan drugs are now being assessed. The Zorginstituut Nederland (ZIN) advises the MoH on the addition of drugs to the basic healthcare package and uses cost-effectiveness criteria to assess them. If entered into the lock, an assessment must be done by the ZIN to decide whether to reimburse the drug, during which time the drug is not included in the approved care package. A study of the first 8 drugs placed in the lock found assessment took on average 9 months, thus delaying market access. A drug is put into the lock if:
- The costs for the treatment of 1 or more new indications throughout the Netherlands are more than €40 million per year. In this case, all new indications are placed in the lock
- The cost for a new indication per patient is €50,000 or more per year and amounts to a total of €10 million or more per year. In this case, only the new indication is placed in the lock
The minister decides which medicines will be placed in the lock on the basis of data from the Horizon Scan for Medicines. Horizon scanning is carried out by ZIN and the expected cost per patient and patient volume are estimates based on data in the public domain. Of the 21 medicines placed in the lock as of February 8, 2022, more than half (n=12; 57%) were orphan drugs. The annual per-patient costs for these 12 drugs, reported by Dutch horizon scanning, ranged from €40,000 to €3,000,000 and annual patient numbers from 1–5 to <386 (see Table 1).
Table 1. ZIN horizon scanning data for orphan drugs in the lock
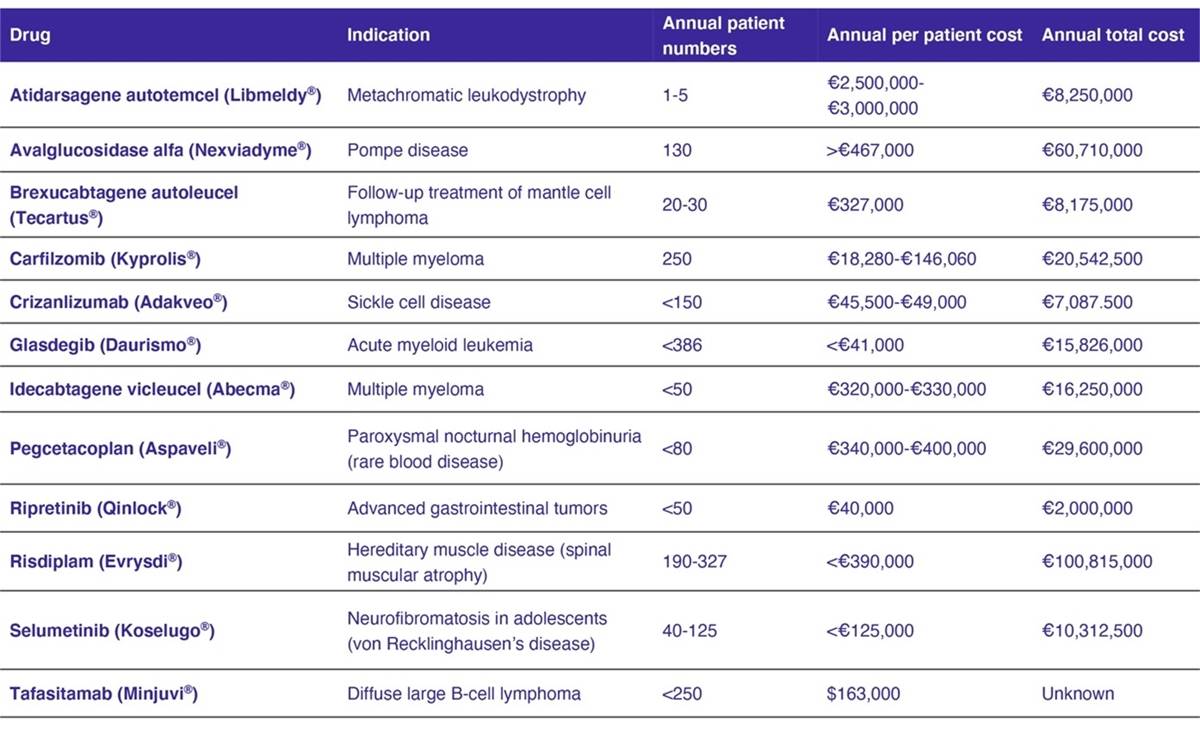
Source: Zorginstituut Nederland. 2022b.
Note the database is live; products are only listed whilst in the lock. Once assessed, they are removed. ZIN states that the tafasitamab patient cost is based on the US price as prices for the Netherlands are still unknown and that ripretinib is estimated to cost €40,000 per patient, assuming that it would be priced similar to sunitinib. These assumptions may or may not turn out to be correct. Crizanlizumab is estimated at €45,000 to €49,000 per patient and, like ripretinib, should not be in the lock per the criteria stated above. Given both were placed in the lock, ZIN may be factoring in potentially higher prices. Indeed, ZIN states crizanlizumab has been placed in the lock until a financial arrangement has been agreed upon, implying the lock is being used as a mechanism to stimulate pricing negotiations.
As treatments for rare cancer forms and blood diseases are frequently represented, we analyzed orphan drugs approved by the EMA in these indications (anatomical therapeutic classification [ATC] codes L01X and B06A). Since 2018, 64% (9/14) of the orphan drugs approved for these indications were placed in the lock. Two of the drugs have dropped out of the lock but have not been reimbursed following assessment by the ZIN, three drugs are now reimbursed, and the rest are currently still in the lock.
Like the Netherlands, the UK National Institute for Health and Care Excellence (NICE) uses cost-effectiveness criteria to recommend the prescription of drugs on the NHS in England and Wales. An incremental cost-effectiveness ratio threshold of £20,000 to £30,000 per quality-adjusted life-year is set for most medicines; however, for highly specialized technology (HST) evaluations, the ICER threshold is increased to £100,000. To meet HST criteria, technologies must target a “small distinct patient group.” Small is not defined, but in practice most orphan medicines do not meet HST criteria; only 17 appraisals have been published since 2015. Since 2018 over 60% (9/14) of orphan drugs for ATC codes L01X and B06A gained a positive recommendation from NICE. None of them were assessed via the HST process, and all of them have either a confidential commercial agreement to bring the incremental cost-effectiveness ratio down below the threshold or are part of a managed access scheme that is likely to include a price discount. This suggests the incremental cost-effectiveness ratio thresholds are being used as a tool to lower prices, similar to the Dutch lock system.
In Germany, the Federal Joint Committee (G-BA), Gemeinsamer Bundesausschuss assesses the benefit provided by new medicines based on clinical data and costs of the product compared with an appropriate comparator set by G-BA. This level of added benefit is then used in pricing negotiations with the National Association of Statutory Health Insurance Funds (GKV-SV). For orphan drugs, additional therapeutic benefit is assumed, and no comparative data is required but the G-BA still assesses the magnitude of the additional therapeutic benefit in order to create the basis for price negotiations. If annual expenditure exceeds €50 million, orphan drugs are evaluated in the same manner as all other drugs and prices renegotiated. For the analyzed L01X and B06A orphan drug sample, the G-BA determined a non-quantifiable additional benefit in 64% (9/14) of the cases, the lowest possible rating for orphan drugs, because the scientific data did not permit a quantification. Still, a non-quantifiable additional benefit provides a positive basis for the price negotiation, as by law, drugs assessed as providing no additional benefit should not have higher annual therapy costs than the appropriate comparator.
As our analyses show, orphan drugs are regularly placed in the Dutch lock. The rules on entry to the lock are not always adhered to and drugs that appear to fall outside the lock criteria according to publicly available cost and epidemiology data may still be placed in the lock pending ZIN assessment. So, orphan drugs do not automatically have an easier route to achieving reimbursement at the desired prices; small population sizes for orphan diseases confer little advantage when it comes to HTA; rigorous evidence is still required, and products are subject to complex access schemes. Clinical development plans need to take these access requirements into account; otherwise, high discounts or worse, refusal to reimburse, might be waiting at the end of the road.
Watts-James J, Konstanski M, Schepis Martinez J. Orphan drugs: trapped in the lock? A review of the lock system in the Netherlands. HTA Quarterly. Summer 2022. https://www.xcenda.com/insights/htaq-summer-2022-orphan-drugs-trapped-in-the-lock
Sources:
- Clarke S, Ellis M, Brownrigg J. The impact of rarity in NICE’s health technology appraisals. Orphanet J Rare Dis. 2021;16:218.
- EMA. 2022a. European public assessment reports. Accessed February 4, 2022. https://www.ema.europa.eu/en/medicines/download-medicine-data#european-public-assessment-reports-(epar)-section
- EMA. 2022b. Rare disease (orphan) designations. Accessed February 4, 2022. https://www.ema.europa.eu/en/medicines/download-medicine-data#rare-disease-(orphan)-designations-section
- G-BA. The benefit assessment of medicinal products in accordance with the German Social Code, Book Five (SGB V), section 35a. Accessed February 4, 2022. https://www.g-ba.de/english/benefitassessment/
- Harari S. Why we should care about ultra-rare disease. Eur Respir Rev. 2016;25:101-103.
- NICE. 2022a. NICE health technology evaluations: the manual. Accessed March 15, 2022. https://www.nice.org.uk/process/pmg36/chapter/economic-evaluation
- NICE. 2022b. Interim process and methods of the highly specialised technologies programme. Accessed March 15, 2022. https://www.nice.org.uk/Media/Default/About/what-we-do/NICE-guidance/NICE-highly-specialised-technologies-guidance/HST-interim-methods-process-guide-may-17.pdf
- NICE. 2022c. Guidance, NICE advice and quality standards. Accessed March 15, 2022. https://www.nice.org.uk/guidance/published?ngt=Highly specialised technologies guidance&ndt=Guidance
- OECD 2018. Pharmaceutical reimbursement and pricing in Germany. Accessed March 15, 2022. https://www.oecd.org/els/health-systems/Pharmaceutical-Reimbursement-and-Pricing-in-Germany.pdf
- HAI (Health Action International). Report: New and affordable medicines in the Netherlands. Accessed March 15, 2022. https://haiweb.org/wp-content/uploads/2018/12/NL-Government-Commitments-on-New-Affordable-Medicines.pdf
- Trip AM, van Steen C, Hensen M. The introduction and implications of the cost-containment measure ‘the sluice’ For expensive innovative drugs in the Netherlands. Value Health. 2018;21(suppl 3):S191.
- Wakap SN, Lambert DM, Olry A, et al. Estimating cumulative point prevalence of rare diseases: analysis of the Orphanet database. Eur J Hum Genet. 2020;28:165-173.
- Zorginstituut Nederland. 2022a. Sluis voor dure geneesmiddelen. Accessed February 8, 2022. https://www.zorginstituutnederland.nl/over-ons/programmas-en-samenwerkingsverbanden/horizonscan-geneesmiddelen/sluis-voor-dure-geneesmiddelen
- Zorginstituut Nederland. 2022b. Horizonscan geneesmiddelen. Accessed February 8, 2022. https://www.horizonscangeneesmiddelen.nl/
- Zorginstituut Nederland. 2022c. Crizanlizumab. Accessed March 15, 2022. https://www.horizonscangeneesmiddelen.nl/geneesmiddelen/crizanlizumab-cardiovasculaire-aandoeningen-overige_niet-oncologische_hematologische_medicatie/versie6




